Last week we got to the stage of completing the manufacture of the tablets in bulk. Now we have to prepare the tablets for sale which first means putting them into bottles.
The bottling process is a pretty straightforward one. The bottles are supplied to us in two parts. One part being the body of the bottle itself and the other being the cap and tamper ring. The tops are tipped into a sorting machine and the bottles are arranged at start of the bottling line. The tablets, soft gels or capsules are tipped into a hopper and then fed down a vibrating stainless chute.
The tablets or capsules pass through counting sensors which ensure that each bottle contains the correct number of tablets or capsules. These sensors are very accurate but occasionally they may count 1 tablet as two. We test the counter accuracy on a regular basis, but occasionally an incomplete tablet can sneak through. We compensate by setting the counter to always add 1 extra tablet/capsule…
So, if you count the number of tablets/capsules in your bottle, you will probably have one extra…and yes, some people do count them.

Photos of the Bottling Line
After the bottling and labelling is complete the batch is quarantined whilst further tests are carried out.
These additional tests are similar to those that we have previously carried out on the raw ingredients…namely microbiological.
Some of these tests are over and above what is required for GMP compliance. They are part of our internal quality system and provide us with peace of mind that everything is in order before product is released to our customers. Although it is unlikely, there is always the possibility of contamination at some point during production, so by doing this we can be 100% confident that we are supplying a pure, safe product.
After these tests are completed and the results confirm that the product has passed all testing, the Quality Assurance team release it from our factory to our distribution department.
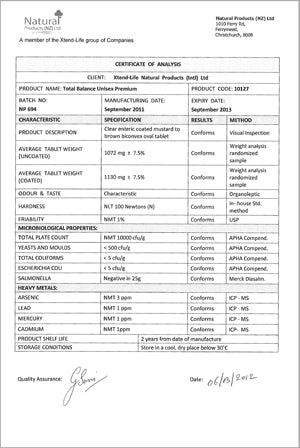
Example of a COA
The distribution warehouse is set up so that all of the previous batch is shipped out to customers before the latest batch is used. This ensures that customers always get fresh product. Also, we can track which customers received which batch.
OK…that completes this little series of how we manufacture our supplements. Of course the focus of this series was the tablets. At a later time we will go through the process for the manufacture of the soft gels for our fish oil products.
Also, if we get expressions of interest from readers wishing to know how we manufacture our skin care products I will write a series on that as well. There are some similarities in that we apply many of the same principles when producing our skin care products as we do for our supplements.
In that respect we differ from many skin care manufacturers who are only loosely regulated. However, we take the position from a nutritional point of view in that the skin is the largest organ in the body and you are indirectly ingesting the ingredients in the skin care products. Therefore they should be manufactured with the same care and attention to safety and hygiene.
I hope that these series of articles have helped gain a better insight in what goes into the supplements that you and I take every day.


 Supplements
Supplements Skincare
Skincare Superfoods
Superfoods Bundles
Bundles












Leave a comment (all fields required)